Carbohydrates Definition
The term “carbohydrates” is applied to a large number of relatively heterogeneous compounds found in all animals and plants. Carbohydrates are polyhydric alcohols with potentially active carbonyl groups which may either be aldehyde or ketone groups. They also include those components which yield them on hydrolysis. Some carbohydrates also contain N and S. Carbohydrates are also called saccharides. Carbohydrates Formula Name of carbohydrate is derived from the fact that the first compounds of this group which were studied had an empirical formula Cx (H2O) y. But now it is known that many carbohydrates contain carbon and hydrogen not in the same proportion as in water. E.g., deoxyribose which is C5H10O4. Classification of Carbohydrates described below.

Classification of Carbohydrates
There is no single satisfactory classification of carbohydrates. One commonly described classification is as follows:-
- Monosaccharides
- Oligosaccharides
- Polysaccharides
- Derived carbohydrates
Flowchart of Classification of Carbohydrates: 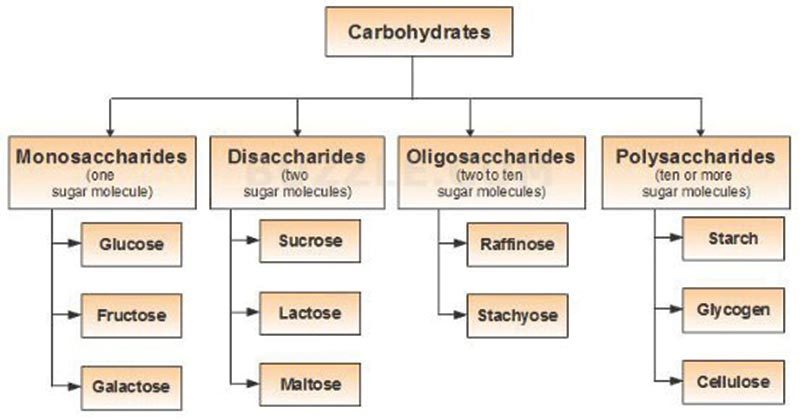
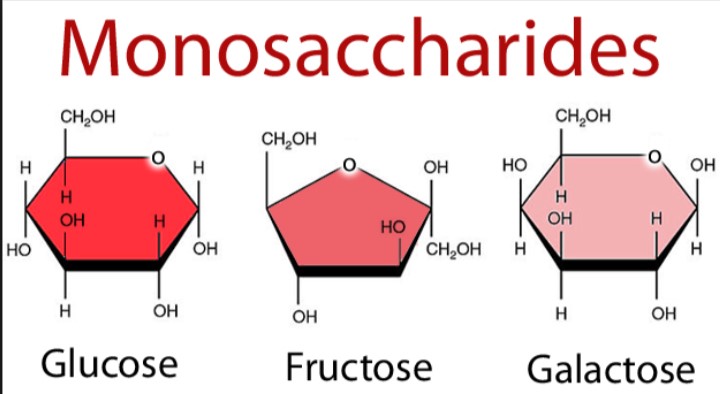
1. Monosaccharides
- Monosaccharides are very simple sugars.
- They cannot be further hydrolyzed into other saccharides.
- Monosaccharides have the empirical formula (CH2O) n where n=3 or some larger number.
- Monosaccharides are either aldoses (containing aldehyde groups) or ketoses (containing ketone groups).
- They are White crystalline sugar solids.
- They have a high tendency to dissolve in water.
- Most have a sweet taste.
A few examples are given below:
- The type of carbonyl group (aldehyde or ketone) and the generic name are combined in naming the monosaccharides. E.g., glucose is an aldose as well as hexose; it is, therefore, an aldohexose. Fructose, on the other hand, is a ketohexose. Glyceraldehyde is an aldotriose.
2. Oligosaccharides
- These on hydrolysis produces 2-10 molecules of monosaccharides sugars.
- In this category, disaccharides are the ones which are physiologically important.
- In oligosaccharides maltose (2 glucose molecule), saccharose or sucrose (1 molecule each of glucose and fructose) and lactose (1 molecule of each glucose and galactose).
- In oligosaccharides, the monosaccharides are joined to each other through glycosidic linkages.
Examples of oligosaccharides: 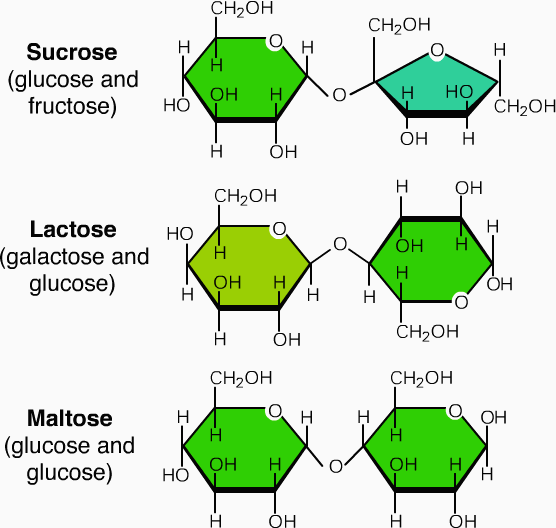
You May Also Like: Meiosis | Phases of Meiosis | Importance of Meiosis
2. Polysaccharides
- These carbohydrates contain a large number of sugar units in their molecules.
- They serve as stores fuel of energy.
- They generally form the basic structural unit of the cell.
Polysaccharides consist of the following types :
- Homopolysaccharides
On hydrolysis, these yields only one type of monosaccharide units These include starch, glycogen, cellulose, and dextrins all of which are glucose polymers.
- Heteropolysaccharides
This is a very large group containing two or more different types of monosaccharide units. These heteropolysaccharides are furtherly divided into the following three subgroups:
- Mucopolysaccharides:
These include hyaluronic acid, heparin, chondroitin sulfates, blood group polysaccharides, and serum mucoids. These substances, in addition to containing carbohydrate parts, also contain acid groups. 2. Mucilages: These include agar, vegetable gums, and pectins; all of these are of plant origin. 3. Hemicellulose: It is also a heteropolysaccharide occurring in the plant kingdom. 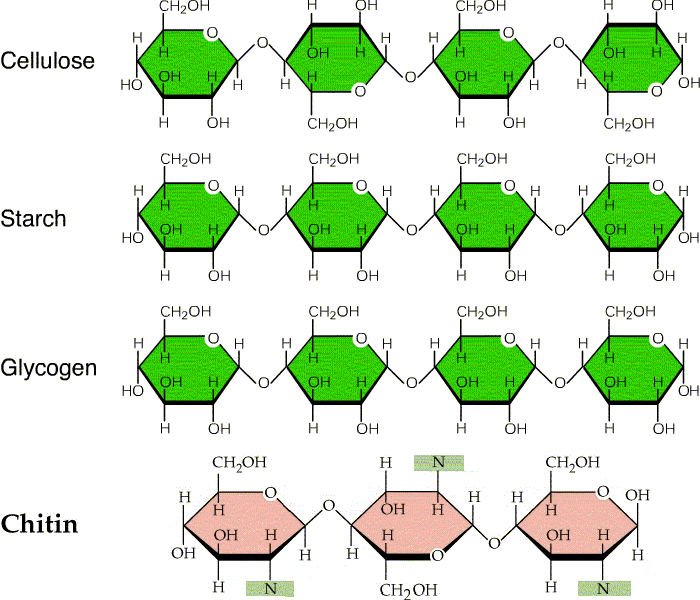
- Derived Carbohydrates:
These type of carbohydrates are usually derived from other carbohydrates by various chemical reactions. These include the following:
- Oxidation producers: Various sugar acids e.g., gluconic acid, glucuronic acid, and glucaric acid, are derived from glucose on its oxidation. Ascorbic acid (vitamin C) is also an oxidation product of glucose and is produced in the bodies of many animals but not in man.
- Reduction products: These are polyhydroxy alcohols, e.g., glycerol and ribitol derived from glyceraldehydes and ribose respectively.
- Amino sugars: These have the NH2 group at C No. 2 and include glucosamine, galactosamine, and mannosamine derived from glucose, galactose and mannose respectively.
- Deoxy sugars: These have fewer oxygen atoms than other sugars. An important example is 2-deoxyribose that is present in the DNA molecule.
- Carbohydrate molecules also are found in combination with proteins and lipids forming glycoproteins, glycolipids, and lipopolysaccharides.
