Carbohydrates
Carbohydrates Definition
The term “carbohydrates” is applied to a large number of relatively heterogeneous compounds found in all animals and plants. Carbohydrates are polyhydric alcohols with potentially active carbonyl groups which may either be aldehyde or ketone groups. They also include those components which yield them on hydrolysis. Some carbohydrates also contain N and S. Carbohydrates are also called saccharides. A polysaccharide is defined as a large molecule composed of many smaller monosaccharides.
Carbohydrates Formula
The name of carbohydrate is derived from the fact that the first compounds of this group which were studied had an empirical formula Cx (H2O) y. But now it is known that many carbohydrates contain carbon and hydrogen not in the same proportion as in water. E.g., deoxyribose which is C5H10O4.

Classification of Carbohydrates
There is no single satisfactory classification of carbohydrates. One commonly described classification is as follows:-
- Monosaccharides
- Oligosaccharides
- Polysaccharides
- Derived carbohydrates
Flowchart of Classification of Carbohydrates: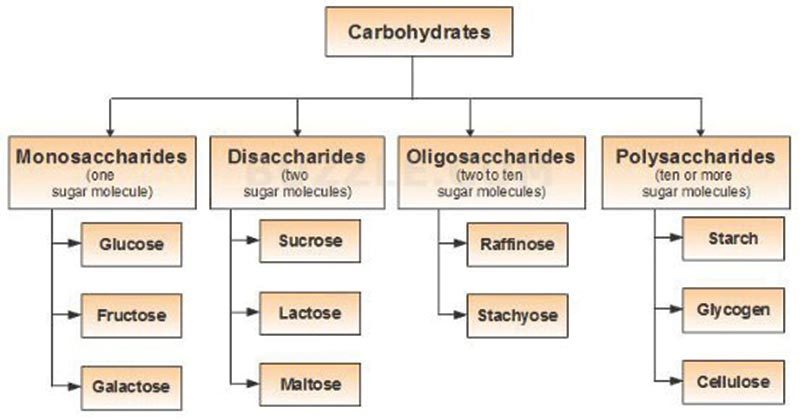
1. Monosaccharides
- Monosaccharides are very simple sugars.
- They cannot be further hydrolyzed into other saccharides.
- Monosaccharides have the empirical formula (CH2O) n where n=3 or some larger number.
- Monosaccharides are either aldoses (containing aldehyde groups) or ketoses (containing ketone groups).
- They are White crystalline sugar solids.
- They have a high tendency to dissolve in water.
- Most have a sweet taste.
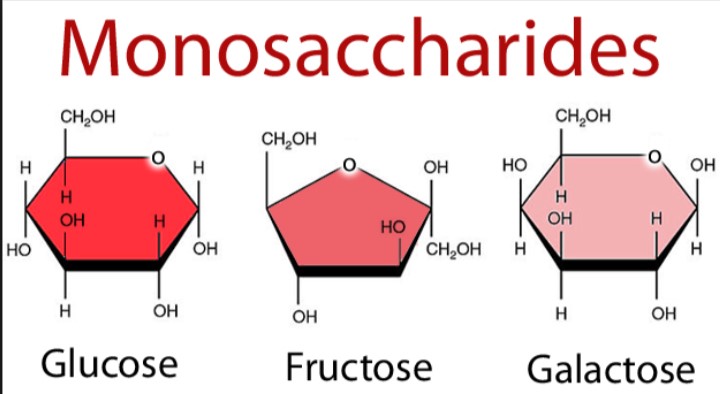
A few examples are given below: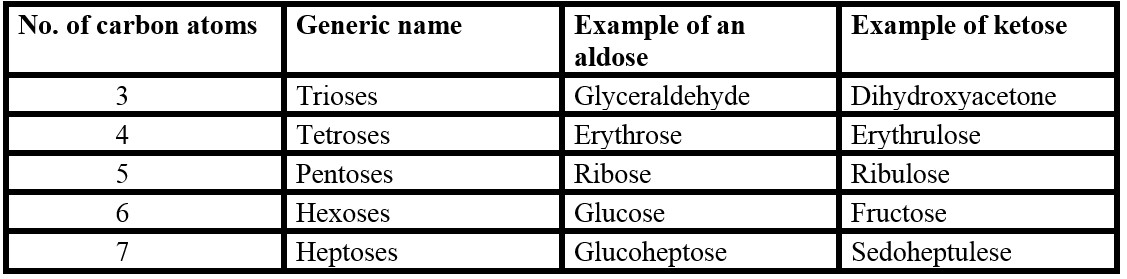
- The type of carbonyl group (aldehyde or ketone) and the generic name are combined in naming the monosaccharides. E.g., glucose is an aldose as well as hexose; it is, therefore, an aldohexose. Fructose, on the other hand, is a ketohexose. Glyceraldehyde is an aldotriose.
2. Oligosaccharides
- These on hydrolysis produces 2-10 molecules of monosaccharides sugars.
- In this category, disaccharides are the ones which are physiologically important.
- In oligosaccharides maltose (2 glucose molecule), saccharose or sucrose (1 molecule each of glucose and fructose) and lactose (1 molecule of each of glucose and galactose).
- In oligosaccharides, the monosaccharides are joined to each other through glycosidic linkages.
Examples of oligosaccharides: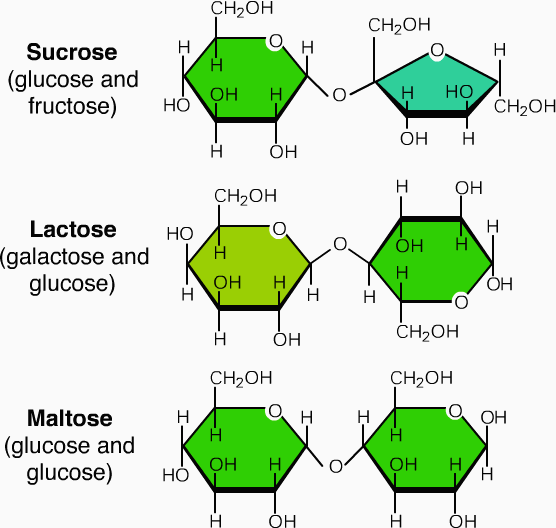
Polysaccharide Definition:
A polysaccharide is defined as a large molecule composed of many smaller monosaccharides. The monosaccharides are simple sugars such as glucose. These small monomers are bind by some special enzymes together and create large sugar polymers or polysaccharides, and are also known as glycan. The polysaccharides can be homopolysaccharide, if all monosaccharides are the same, while if all monosaccharides are varying, it is said to be heteropolysaccharide. The polysaccharides take on a form variety, on the basis of which monosaccharides are connected and which carbons are connected in the monosaccharides. In a straight chain of monosaccharide, a molecule is known as linear polysaccharide, and a chain consisting of arms and turns is called a branched polysaccharide.
Functions of a Polysaccharide:
The polysaccharides can perform various functions in nature on the basis of their structure. Some polysaccharides are used in order to store energy, some used for sending cellular messages and many others used to support the cells and tissues.
- Storage of Energy:
Many polysaccharides perform the function of the storage of energy in an individual. The enzymes which generate energy only work on monosaccharides that are stored in the polysaccharide. The polysaccharides typically fold each other may consist of many monosaccharides in the dense regions.
The polysaccharides are not only used for store energy but they also permit for change in a concentration gradient that may influence cellular uptake of nutrients and the water.
- Cellular Communication:
When some of the polysaccharides are made a covalent bond with proteins or lipids, they become Glycoconjugates. Glycolipids and glycoproteins are used for sending signals between and in the cells. The proteins are referred to as particular organelle and used in specific polysaccharides which help the cell move it to certain organelle.
The polysaccharides are also referred to as special protein that helps in binding the proteins, vesicles, or the other substances to microtubules.
- Cellular Support:
The polysaccharides also perform the function of providing support to the cells. The polysaccharides cellulose used to support all the plants on the earth. Other organisms including insects and fungi use chitin in order to support the extracellular matrix in the rounding of their cells. A polysaccharide may be mixed with any of the other components in order to produce tissues that are much rigid, less rigid or even has special material properties. By simply rearranging of structure, the polysaccharides can become molecules storage to stronger fibrous molecules.

Structure of a Polysaccharide:
All the polysaccharides are created by the basic process that is the same for all. Firstly, the monosaccharides connect via glycosidic bonds. The monosaccharides are called residues when they are in polysaccharides.
The structure of a combination of molecules determines the structure and the properties which are the result of polysaccharide. The complex interaction lies between their hydroxyl groups (OH), the other sides of molecules configure the molecules.
The glycosidic bonds between the monosaccharides contain one molecule of oxygen with bringing two carbon rings. The bond is created when a hydroxyl group lost from one carbon molecule and hydrogen loss from the hydroxyl group of another monosaccharide.
Examples of a Polysaccharide:
Examples of polysaccharide are mention below:
- Cellulose and Chitin:
Both the cellulose and chitin are structural polysaccharides that contain many thousand glucose monomers are combining in long fibers. The difference lies between these two structural polysaccharides are only that the side chains attach within the carbon rings of monosaccharides.
In chitin, the glucose monosaccharides modify by a group consists of more carbon, nitrogen, and oxygen. The side chain produces a dipole that increases the bonding of hydrogen. On the other hand, the cellulose may produce hard structures such as wood, but chitin can produce harder structures such as shell, limestone, and marble when it compressed.
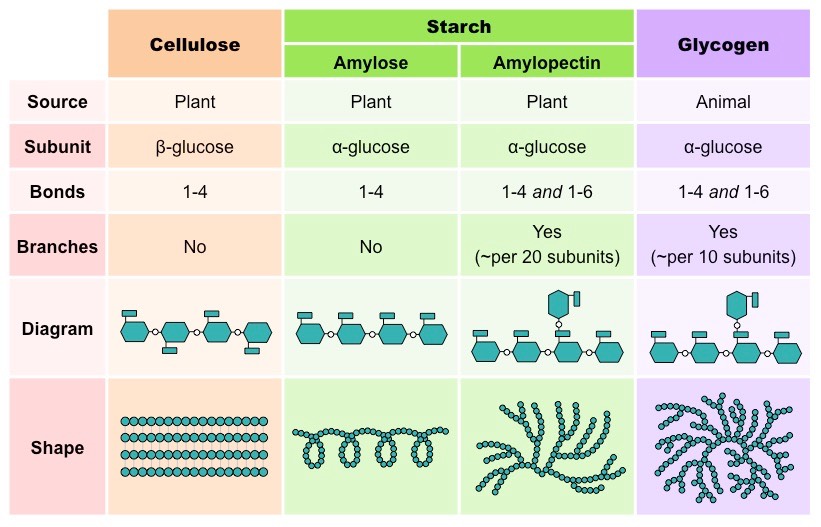
- Glycogen and Starch:
Glycogen and starch are the most important polysaccharides used for storage, are produced by animals and plants. These polysaccharides form from the central starting point and spiral outward due to their complex patterns of branching. The large branch molecule creates Granules or cluster with the help of many proteins which are attached to the individual polysaccharides. When glycogen and starch molecule breaks down, the enzymes are responsible to start at the ends from the center.