Acetic Acid | Definition, Formula & Uses
Definition of Acetic acid
What is Acetic Acid: It is a fully symmetrical molecule. The name of acetic acid comes from acetum, which is Latin for vinegar. The acid does not give rise to a sour taste in any significant amount. It is the product of microbial fermentation. The sour taste is actually because of lactic acid, which gives rise to full mouth-puckering sourness. Glacial acetic acid is basically the anhydrous form of acetic acid, which is undiluted or free of water form.
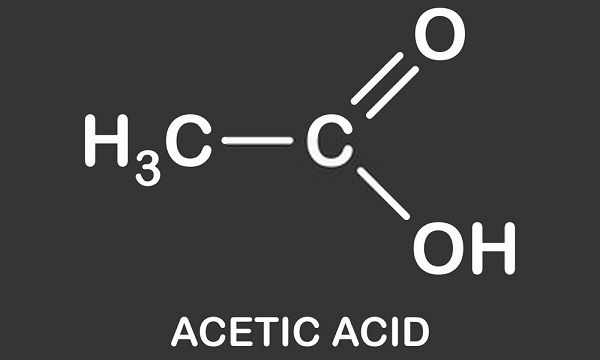
Formula Of Acetic Acid
It is also known as ethanoic acid, and it has the chemical formula CH 3 COOH. The acetic acid formula is showed below:
The molar mass of acetic acid
Acetic acid pKa value
How Is Acetic Acid Made?
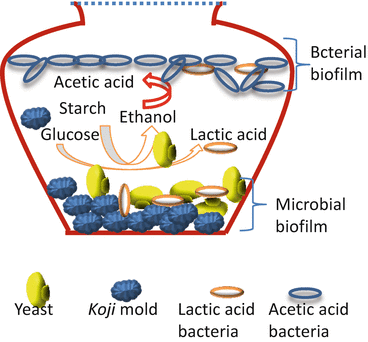
The bacteria used to make acetic acid are called Acetogenins. They use anaerobic respiration and can reduce carbon dioxide into carboxylic acids, like the glucose shown below:
Glucose (C 6 H 12 O 6) + 2 CO₂ → 2 CH 3COOH
The process is very similar to the conversion of the starches in corn into ethanol. The fermentation process can take place at 80°C and under high pressure when using Zymomonas bacteria. They break down sugar to produce electricity and hydrogen gas, which can be used as a fuel source. In contrast, other bacteria, such as Moorella thermoacetica can generate acetic acid from carbon monoxide.
Reactions of Acetic acid
The reaction of acetic acid, also called ethanoic acid, with atmospheric oxygen, happens in two steps.
A single electron is transferred from a hydrogen atom within acetic acid to an oxygen molecule in the first step. The reaction is reversible, and the second reaction below will then take place, which will give water and carbon dioxide as products:
CH 3 COOH + O 2 ⇌ CH 3 COO- + H+ + HO 2 ¯
The second step is an irreversible formation , which takes place in the cytoplasmic membrane. The electron transfer takes place via a dehydrogenase enzyme, which is also called the Krebs cycle. The following equation shows the formation of acetic acid:
Acetyl Co-A + 3 NAD+ ⇌ 2 NADH + CO 2 +CH 3 COOH
A single hydrogen atom is removed from acetyl Co-A and moves to one of the three nitrogen atoms in the enzyme complex. The movement pushes electrons out into cytochrome and generates a flow of electric current, and thus energy is released and used by the cell. The acetyl Co-A returns to the mitochondria, providing carbon atoms for use in the biosynthesis of the fatty acid synthesis process.
As soon as the electrons are removed from acid, water molecules attach themselves and keep them away from the acetyl Co-A molecule. The result is the formation of water and acetic acid.
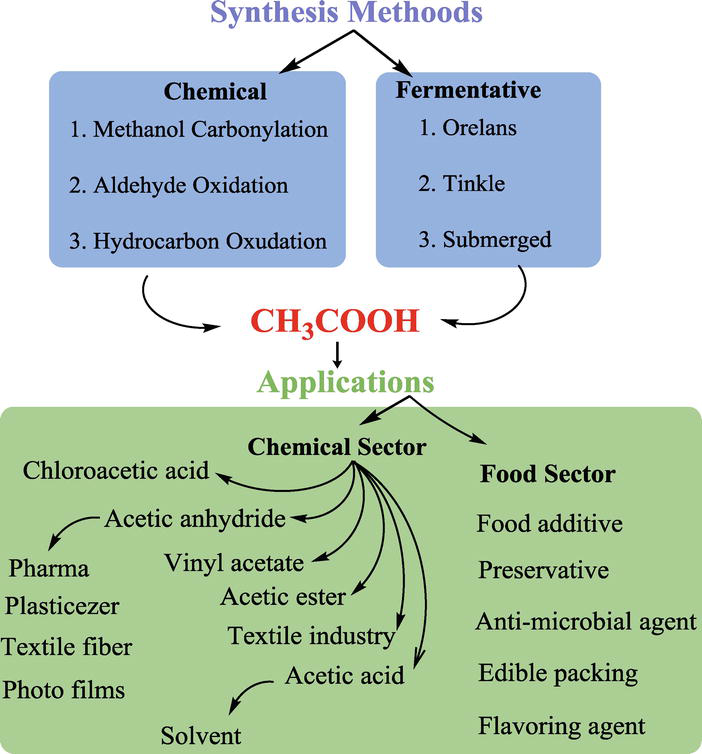
What is acetic acid used for?
Uses of Acetic Acid are described below:
- It has many functions in nature because it can act as an energy carrier, a chemical building block for biological molecules, is used to manufacture cosmetics and drugs, and is a component of Kreb’s cycle that generates energy in living organisms.
- It can be stored as an ester, and it is a precursor for amino acid synthesis.
- It also plays a vital role in other areas, such as how it acts as an antibacterial agent, functions as a preservative, eliminates unwanted microbes and is used as an antiseptic.
- It has also proved to be effective in the treatment of skin conditions such as acne.
- It is not harmful because it can be found in food. However, high concentrations are corrosive and irritating to the eyes, nose, throat, and stomach.
- It is also the principal ingredient in vinegar, which makes it sour.
- Most of the acid produced is used for commercial uses, such as vinyl acetate monomer and cellulose acetate.
- It also finds extensive use in organic synthesis, where it is used as a reagent for the synthesis of acetic anhydride, acetyl chloride, and other organic compounds.
- It is also used to prepare other chemicals used to manufacture drugs, plastics, cosmetics, food additives, and dyes.
- It is also found in many household items, such as perfumes, disinfectants, cosmetics, pickles, vinegar, and glues. The result is that it has many applications across diverse fields.
- It can also be used in the treatment of burns by destroying harmful microbes to prevent infection. It has also proved effective in treating acne when applied topically or used systemically. When taken internally, it acts as a laxative, and it can also treat dysentery and bronchitis.
- It is an excellent preservative, and it prevents the growth of bacteria that cause food to spoil.
- The low pH makes the environment highly acidic, where dangerous microbes cannot survive, causing natural fermentation. This process provides a natural treatment for many ailments, such as those of the stomach and intestines.
- It is also a good preservative for natural perfumes because it can slow down or prevent microbial metabolisms. This prevents chemical reactions that break down the essential oils present in the aroma, which eventually causes it to decay.